LPS - Pulmonary Inflammation
Discover how Melior’s unique phenotypic screening platforms can uncover the untapped value of your candidate therapeutic
Airway diseases such as chronic pulmonary inflammation (including chronic obstructive pulmonary disease and asthma) exact an ever-increasing toll on human health as the population ages and environmental pollutants (including tobacco smoke) damage lung tissue of young and old alike. These conditions are induced and exacerbated by pro-inflammatory molecules keyed to respond to acute infection, but often result in chronic immunological responses that induce acute crises and damage lung tissue over time.
An important goal in pharmaceutics is to break the inflammatory cascade by blockage or suppression of specific cytokines and chemokines known to induce and amplify lung inflammation. Two such proteins, monocyte chemoattractant protein (MCP-1), and tumor necrosis factor alpha (TNF-α), are found to be associated with early and chronic infection and inflammation in humans, and are mirrored in rodent models of airway inflammation including the LPS mouse model described below.
Intranasal administration of lipopolysaccharide (LPS), a bacterial endotoxin, has been shown to initiate a pro-inflammatory response in the lungs of mice. This LPS-induced inflammation model recapitulates aspects of the inflammatory cascades that are associated with pulmonary inflammation associated with lung disease in humans, and so is useful as a screen for compounds that may disrupt these cascades and attenuate or abort the disease process.
In this study, we used steroidal anti-inflammatory Dexamethasone to pharmacologically validate this model of MCP-1 induction in mice four hours after LPS pulmonary challenge.
Ready to get started or looking for a custom model?
Contact us today for more information about our bespoke research models and to discuss how we can help you answer your unique research questions.
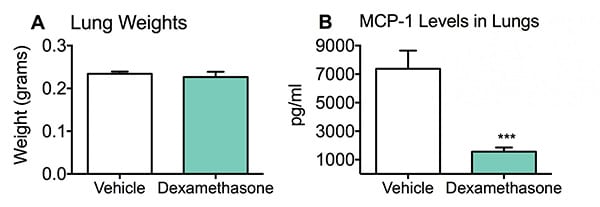
After acute sensitization via intranasal administration of LPS, mice were positioned to provide optimal inhalation and passive gravitational flow of LPS directly to the lungs for approximately five minutes to allow the fullest penetration of LPS to the lungs before awakening. Four hours after LPS challenge, lungs were dissected, collected and processed for MCP-1 ELISA analyses. Dissected lungs from vehicle and Dexamethasone groups resulted in similar weights (A); however, Dexamethasone administration significantly suppressed levels of MCP-1 in the lungs compared to vehicle treated mice (B). Data are mean ± SEM; ***p<0.001 compared to vehicle (N=8).
Dexamethasone was able to significantly reduce the levels of MCP-1 in lung tissue four hours after LPS challenge. This LPS model stands as a good predictor of pulmonary anti-inflammatory compounds. As a screen, it can point the way for further studies of promising compounds, in viewing relative levels of cytokine MCP-1.
Frequently Asked Questions
Lipopolysaccharide (LPS) is the major component of gram-negative bacteria cell walls and can cause an acute inflammatory response by triggering the release of a vast number of inflammatory cytokines in various cell types. LPS is widely recognized as a potent activator of monocytes and macrophages.
Absolutely! If you know your test agent targets a specific set of cytokines, we can test for the up/down regulation of those cytokines using ELISA kits. Make sure to talk to our scientists to ensure your cytokine of interest is involved in this inflammation model.
Synonyms: COPD, asthma



 Interested in running a Pulmonary Inflammation study?
Interested in running a Pulmonary Inflammation study?